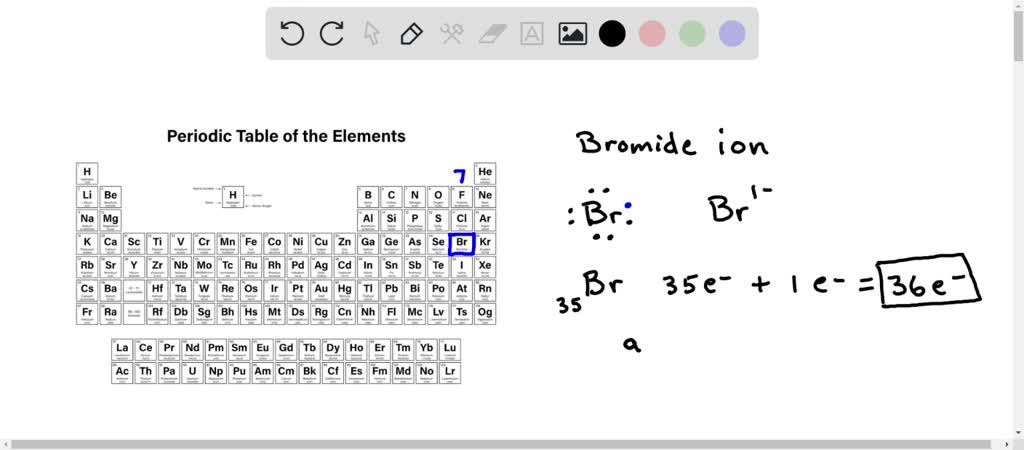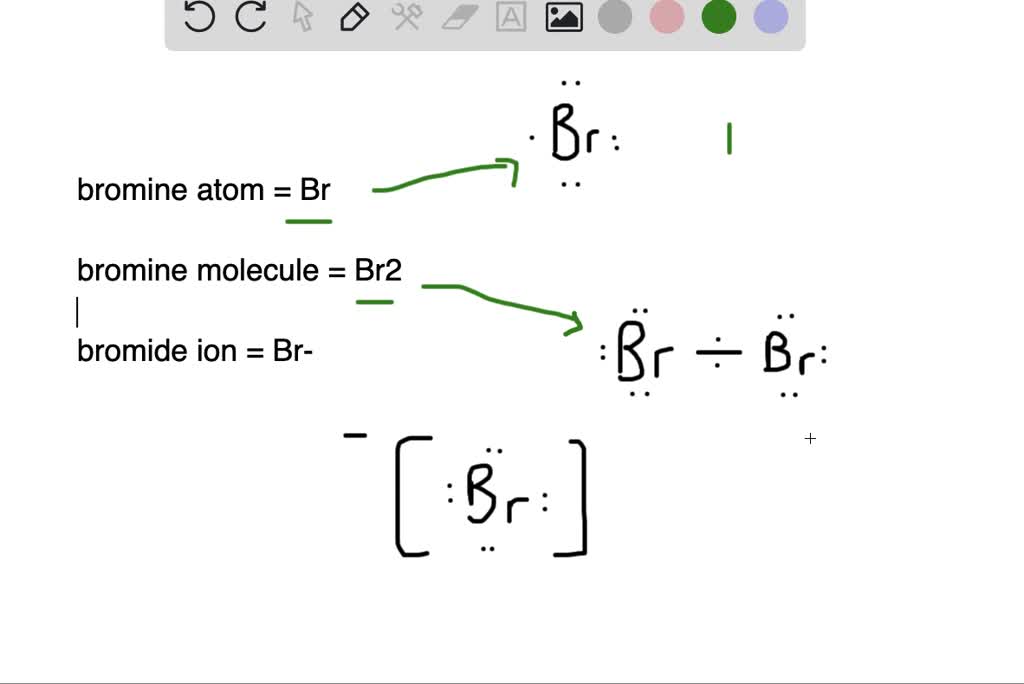
SOLVED:What is the difference between (a) a bromine atom, (b) a bromine molecule, and (c) a bromide ion? Draw the Lewis structure for each.

mass spectrum of 2-bromobutane C4H9Br CH3CHBrCH2CH3 fragmentation pattern of m/z m/e ions for analysis
33. Br ions form a close packed structure. If the radius of Br ion is 195pm, calculate the radius of the cation that just fits into the tetrahedral hole. Can a cation
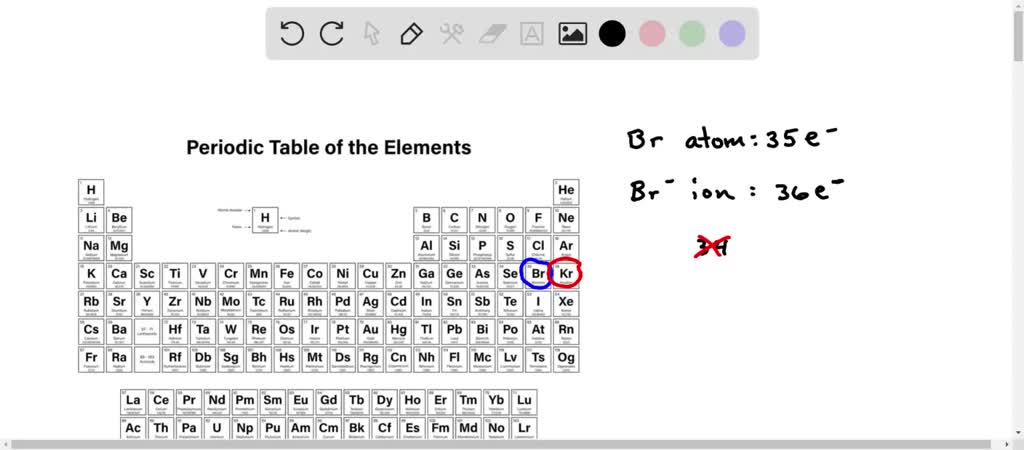
SOLVED: Bromine tends to form simple ions which have the electronic configuration of a noble gas. What is the electronic configuration of the noble gas which the bromide ion mimics? Group of

Why is potassium and bromide KBr? Why does Br not follow 2n2? Br = 35 electrons, 2, 8, 18, 7. Why does the shell not fill to 32 and is happy with

The Multiple Role of Bromide Ion in PPCPs Degradation under UV/Chlorine Treatment | Environmental Science & Technology
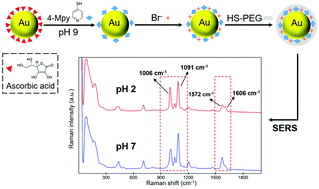
Bromide ion-functionalized nanoprobes for sensitive and reliable pH measurement by surface-enhanced Raman spectroscopy - Analyst (RSC Publishing)

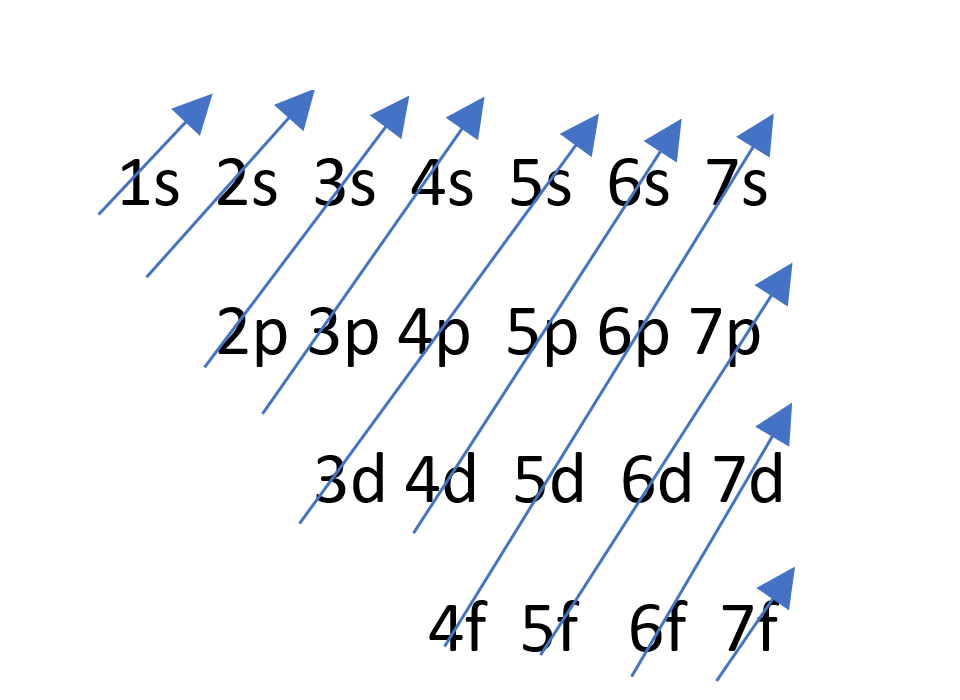


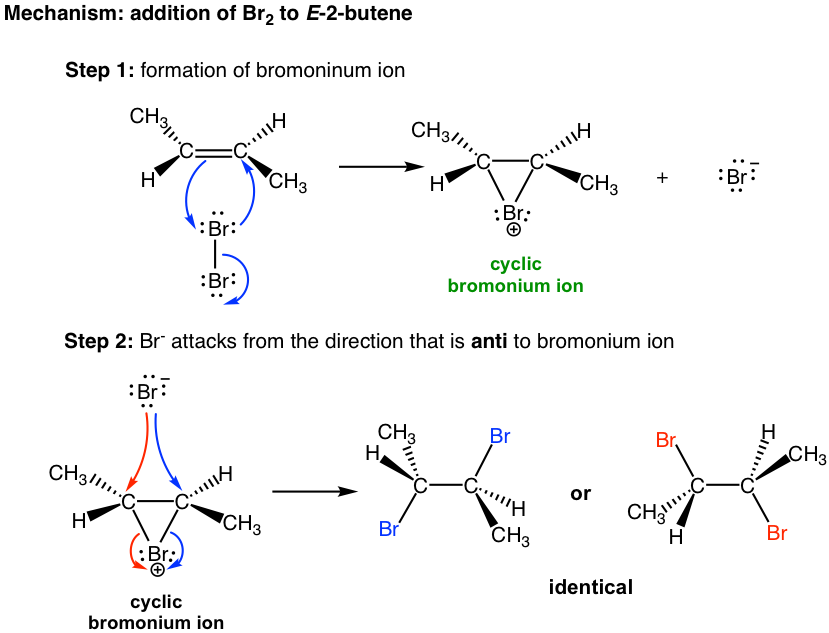
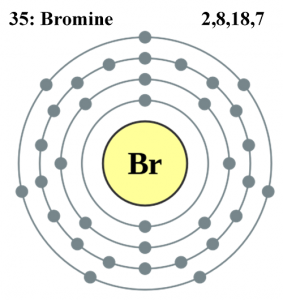






.jpg)